This post is also available in: 简体中文 (Chinese (Simplified))
Hydrogen is already part of the reduction-gas mix in mainstream direct-reduction processes—alongside carbon monoxide, of course. Most notably, perhaps, the Finmet (Finored) process, introduced by Primetals Technologies (VAI) in the 1990s, uses more than six parts of hydrogen for every part of carbon monoxide.
This flexibility of direct-reduction processes provides an enticing way for plant owners to switch to hydrogen in a gradual fashion, increasing the addition of hydrogen over time as prices come down. It also underlines the value of new investment into direct-reduction plants: Whatever the future might hold in terms of emissions regulations or prices for raw materials, direct-reduction technology allows for maximum adaptability.
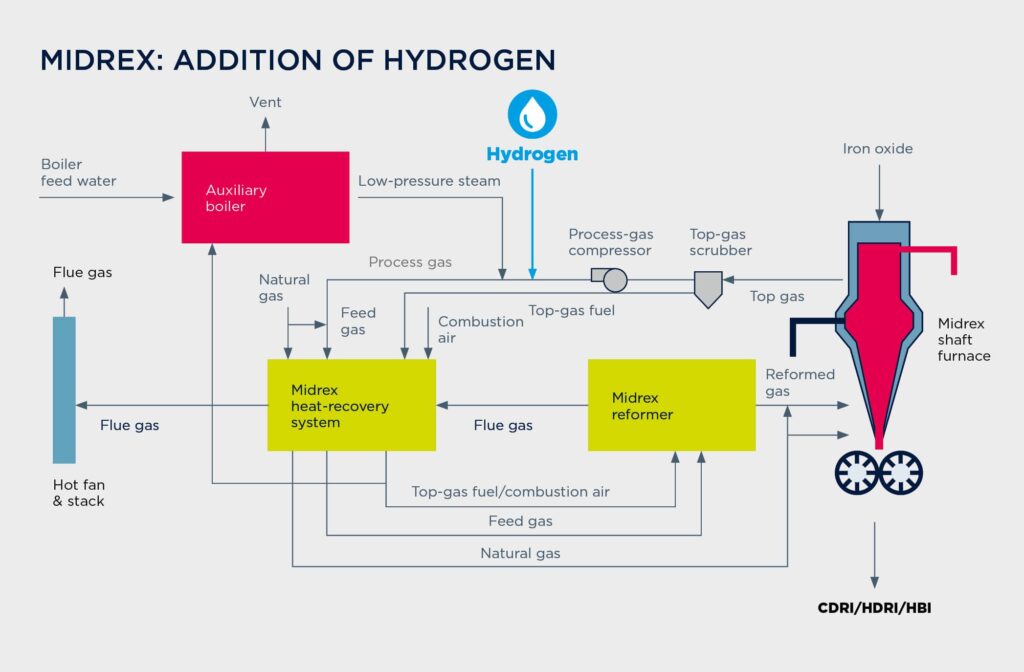
Adding hydrogen to the Midrex process
Midrex direct-reduction plants produce roughly 60 percent of the world’s DRI and have been available from Primetals Technologies for more than 30 years. The reducing gas—mainly a mixture of H2 and CO—is produced from natural gas in a special CO2 reformer supplied by Midrex Technologies. Without any modifications to the equipment, the process allows up to 30 percent of the natural gas to be replaced by hydrogen. To take an example, 60,000 Nm³/h of hydrogen could be brought in to substitute 20,000 Nm³/h of natural gas in the process. With minor additions to the equipment (to protect the reformer), the rate can reach as high as 100 percent—the limit may be determined by the required carbon content in the final product. The process can easily accommodate fluctuations in the hydrogen-addition rate, allowing the plant to react to changing hydrogen supplies (which are to be expected when sourcing the gas from water electrolysis with renewables like wind or solar).

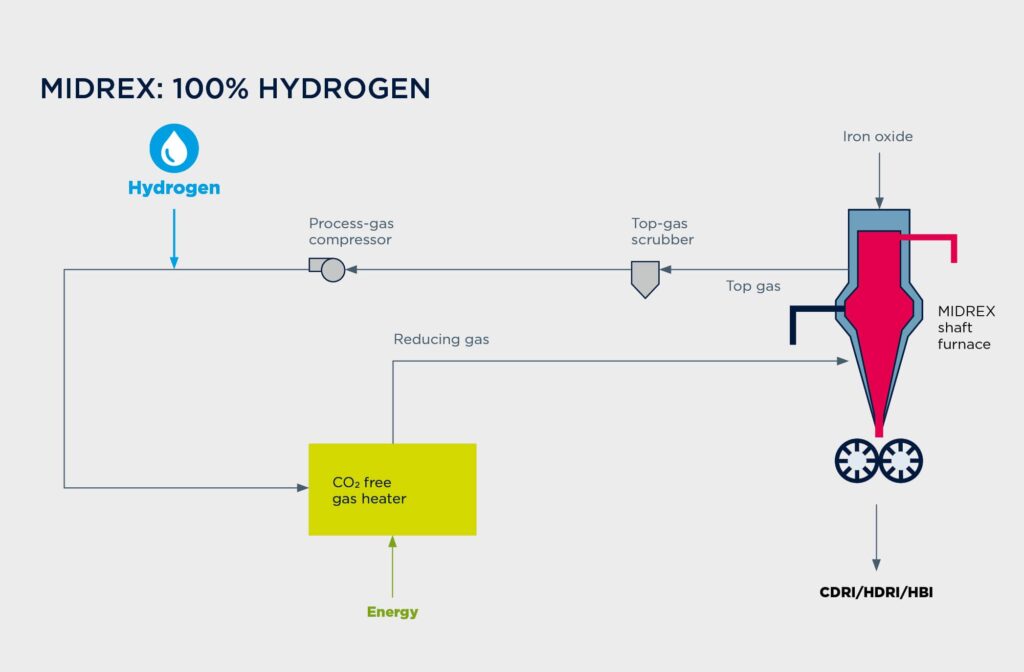
Midrex on 100% Hydrogen
If hydrogen is to be used as the sole reductant permanently, the natural-gas reformer can be replaced by a reduction-gas heater. H2 will be converted to H2O during reduction and condensed in the top-gas scrubber. Since there is no source of carbon monoxide in the process loop, there is no need for a CO2 removal system. The process uses approximately 550 Nm³ of hydrogen per ton of DRI for reduction. Additionally, it requires about 250 Nm³ of H2 per ton of DRI for heat—which can be accommodated by other energy sources.
MORE INFORMATION
Click here to find out more about DRI plants using the MIDREX direct-reduction process.